Free-falling
Snow
... Making snowflakes the natural way... |
|
Another technique for
producing large numbers of snowflakes is to grow them as they fall freely through the air
in a growth chamber. Snowflakes made this way don't grow as large as the natural
variety, however, since our growth chamber is not as large as a cloud. But they are
just the right size for many purposes, and we can photograph them when they fall onto a
window at the bottom of the chamber. |
| Growth
in a Convection Chamber |
 We use a convection chamber to grow these crystals,
shown schematically at right. Basically it is just a cold chamber about a meter
tall, with two containers of heated water on the bottom. Convection mixes the water
vapor into the cold air, creating supersaturated air for growing snowflakes. We
nucleate crystals by dropping a speck of dry ice in the chamber, or by rapidly expanding
some cold compressed air inside the chamber. We use a convection chamber to grow these crystals,
shown schematically at right. Basically it is just a cold chamber about a meter
tall, with two containers of heated water on the bottom. Convection mixes the water
vapor into the cold air, creating supersaturated air for growing snowflakes. We
nucleate crystals by dropping a speck of dry ice in the chamber, or by rapidly expanding
some cold compressed air inside the chamber.
The crystals float until they grow to about 10-100 microns in size, at which
point they fall to the chamber bottom. |
| Crystals
Grown at Different Temperatures |
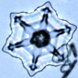
| Growth at -15 C. The most dramatic snow crystal growth
occurs at -15 degrees Celsius and at high supersaturations (>5 percent), which results
in the growth of plates and stellar dendrites (see the Snowflake Primer). |
        |
 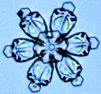       |
 The images above are only a small sample of the crystals we grew using the
free-fall technique; the majority were simply measured and discarded. The first
image shows a crystal with a tip-to-tip diameter of 140 microns, and all the images on
this page are shown at the same scale. The image at right shows a mosaic of snow
crystals grown at -15 C. The images above are only a small sample of the crystals we grew using the
free-fall technique; the majority were simply measured and discarded. The first
image shows a crystal with a tip-to-tip diameter of 140 microns, and all the images on
this page are shown at the same scale. The image at right shows a mosaic of snow
crystals grown at -15 C. |
  These crystals were all grown under quite similar conditions, although some grew for a
longer period of time before falling onto the observation window. The longest growth
time was about two minutes. Even the small variations in temperature and
supersaturation within the growth chamber resulted in the great variety of forms
seen. This demonstrates that the final snow crystal shape is very sensitive to
growth parameters, especially at -15 C when the supersaturation is high.
These crystals were all grown under quite similar conditions, although some grew for a
longer period of time before falling onto the observation window. The longest growth
time was about two minutes. Even the small variations in temperature and
supersaturation within the growth chamber resulted in the great variety of forms
seen. This demonstrates that the final snow crystal shape is very sensitive to
growth parameters, especially at -15 C when the supersaturation is high.
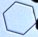
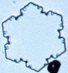
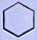
We also grew many snow crystals at lower supersaturation levels (again at -15
C), and under such conditions the crystals nearly always grow as simple hexagonal plates,
similar to the one at left. Next to it is a photograph of a human hair at the same
magnification. |
| |
| Growth at -5 C. Another interesting temperature for snow
crystal growth is at -5 degrees Celsius, which for moderately high supersaturations (about
5 percent) yields hollow column growth (see the Snowflake Primer). |
  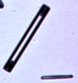    
|
| Some of the smaller crystals grown at -5 C are in the
form of nearly solid (hexagonal) columns, while larger crystals (or those grown at higher
supersaturations) exhibit the distinctive hollowing of the basal faces. This
hollowing is a result of an instability in faceted growth, which causes the protruding
edges to grow faster than the centers of faces; the same instability produces dendritic
arms on plate-like crystals (see Snowflake
Branching). Several of the above crystals also show the twin-prism
morphology, indicating that the initial seed crystal formed with cubic, not hexagonal
symmetry (see Unusual Forms). |
| |
| Growth at -2 C. At -2 degrees Celsius snow crystal
growth becomes plate-like again, although the growth rates are not as high as at -15 C
(see the Snowflake
Primer). Thus the plates are smaller, and they show some qualitative
differences. Note, for example, that the larger crystals (at the far right
below), which grew at the highest supersaturation levels, show distinctive rounded
extensions. These reflect the roughening transition that occurs near the melting
point -- the ice surface becomes microscopically rough, so the crystal boundaries are no
longer faceted. |
        |
|
| The
Hardware |
  At the far right we have a picture of the
growth chamber, which is covered with styrofoam insulation and topped with a
high-intensity lamp. The refrigerator is at the lower left in the image. The
principal players this year -- Audrey Chng, Ken Libbrecht, and Haitao Yu -- are shown in
the near image. At the far right we have a picture of the
growth chamber, which is covered with styrofoam insulation and topped with a
high-intensity lamp. The refrigerator is at the lower left in the image. The
principal players this year -- Audrey Chng, Ken Libbrecht, and Haitao Yu -- are shown in
the near image.
Since the crystals grown in the free-fall chamber were quite small, we used a
high-quality microscope to observe them (N.A. = 0.28). Most of the crystals
photographed above were not much larger than the diameter of a human hair.
In addition to taking pictures, we used this apparatus to make a number of
precise measurements of the growth rates of ice crystals under different conditions. |
