When water freezes into ice, the water
molecules stack together to form a regular crystalline lattice, and the ice lattice has
six-fold symmetry (see the Primer).
It is this hexagonal crystal symmetry that ultimately determines the symmetry of
snow crystals.
But then one must ask how molecular forces, which operate at the molecular
scale to produce the crystal lattice, can control the shape of a snow crystal some ten
million times larger. The answer to this has to do with how crystals form facets.
Facets appear on many growing crystals because some surfaces grow much more
slowly than others. If we imagine beginning with a small round ice crystal, then
mostly we would find that the surface was quite rough on a molecular scale, with lots of
dangling chemical bonds. 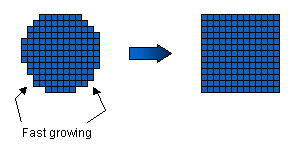 Water molecules from the air can readily attach to these rough surfaces,
which thus grow relatively quickly. The facet planes are special, however, in that
they tend to be smoother on a molecular scale, with fewer dangling bonds. Water
molecules cannot so easily attach to these smooth surfaces, and hence the facet surfaces
advance more slowly. After all the rough surfaces have grown out, what remains are
the slow-moving facet surfaces. The picture at right shows the idea for a crystal
with four-fold symmetry (which is easier to draw). Water molecules from the air can readily attach to these rough surfaces,
which thus grow relatively quickly. The facet planes are special, however, in that
they tend to be smoother on a molecular scale, with fewer dangling bonds. Water
molecules cannot so easily attach to these smooth surfaces, and hence the facet surfaces
advance more slowly. After all the rough surfaces have grown out, what remains are
the slow-moving facet surfaces. The picture at right shows the idea for a crystal
with four-fold symmetry (which is easier to draw).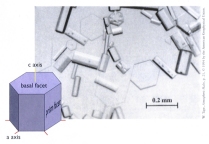 Faceting in snow crystals
produces hexagonal prisms like the ones at left, which are the simplest form of snow
crystals. These specimens were collected at the South Pole by Walter Tape (see Photos),
where the crystals grow very slowly, allowing the facets to fully develop. Faceting in snow crystals
produces hexagonal prisms like the ones at left, which are the simplest form of snow
crystals. These specimens were collected at the South Pole by Walter Tape (see Photos),
where the crystals grow very slowly, allowing the facets to fully develop.
|
