Research
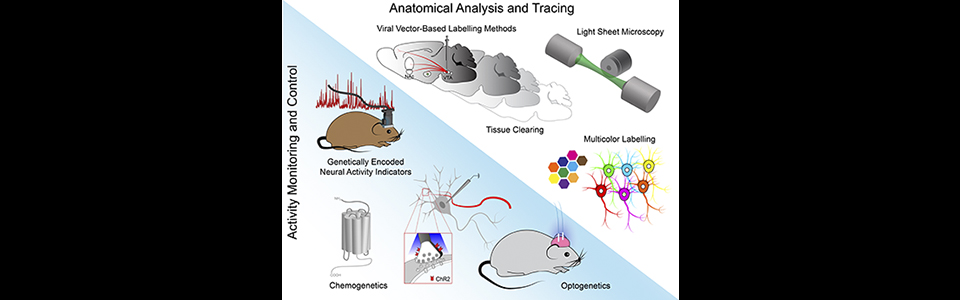
The Gradinaru Lab studies the mechanism of action for deep brain stimulation (DBS), a therapeutical option for motor and mood disorders such as Parkinson’s and depression. Our previous work highlighted the importance of selectively controlling axons and not local cell bodies in modulating behavior, a principle that might play a generalized role across many effective deep brain stimulation paradigms. We are now particularly interested in the long-term effects of DBS on neuronal health, function, and ultimately behavior.
In addition, the lab continues to push forward optogenetic technologies by developing tools for electrical and biochemical control and localizing them to subcellular compartments. To achieve the goals of neuronal circuits investigation and tool development for neuroscience the Gradinaru lab uses advanced Molecular and Synthetic Biology; Electrophysiology (in vitro and in vivo); Behavior; Imaging (2-photon), Optogenetics (gene delivery of photosensitive proteins to specific cell types) and CLARITY (slicing-free whole brain imaging and molecular phenotyping).
Gradinaru Lab is a great fit for any interdisciplinary-minded person. Projects in the lab range from studying the impact of neuromodulation on neurodegeneration and behavior to engineering needed tools (molecular, cellular, hardware) for neuroscience research. If you are interested in joining our team, please email Dr. Gradinaru (viviana at caltech.edu) your CV and a brief description of your scientific interests.
Work in the Gradinaru Laboratory at Caltech is funded by the following awards (to VG): NIH Director's New Innovator IDP20D017782-01;NIH/NIA 1R01AG047664-01; NIH BRAIN1U01NS090577; NIH/NIMH 1R21MH103824-01; Beckman Institute for Optogenetics and CLARITY; Pew Charitable Trust; Kimmel Foundation; Human Frontiers in Science Program; Mallinckrodt Foundation; Gordon and Betty Moore Foundation through Grant GBMF2809 to the Caltech Programmable Molecular Technology Initiative; Michael J. Fox Foundation; Caltech-GIST; Caltech-City of Hope Biomedical Initiative.
Examples from recent work
The Gradinaru Lab reported the first case of whole-body clearing – transparent rodents that can be used to obtain detailed maps of both central and peripheral nerves at their target organs throughout the body (Yang et al., Cell, 2014; Treweek et al., Nature Protocols, 2015) as well as for bone clearing (Greenbaum, Chan et al., Science Trans Med, 2017).
In most recent work (Cho et al., Neuron, 2017), the group has delineated novel arousal-promoting dopaminergic circuits that might be at the root of sleep disturbances common to numerous neuropsychiatric disorders.
To gain real-time feedback from modulated circuits, the group has developed genetically encoded voltage sensors from microbial opsins (Flytzanis, Bedbrook et al., Nature Comm. 2014). To facilitate delivery of such controllers and sensors they developed viral vector screening methods, resulting in capsids capable of crossing the Blood-Brain-Barrier for non-invasive brain-wide transduction in adults after systemic delivery (Deverman et al., Nature Biotech., 2016) and a method for sparse stochastic Golgi-like genetic labeling for morphology assesment (Chan et al., Nature Neurosci., 2017).
To extract functional information from cleared tissue, Gradinaru and collaborators also reported methods for multi-color, multi-RNA imaging in deep in cleared tissue. By using single-molecule hybridization chain reaction (smHCR), tissue hydrogel embedding and clearing, and light-sheet microscopy they detected single-molecule mRNAs in mm-thick brain slices (Shah et al., Development 2016); with rRNA labeling they and collaborators mapped the identity and growth rate of pathogens in cleared clinical samples (DePas et al., mBio, 2016).